Liquefaction of Gases
| Institution | UNIVERSITY |
| Course | BACHELOR OF SCIENCE... |
| Year | 1st Year |
| Semester | Unknown |
| Posted By | stephen oyake rabilo |
| File Type | |
| Pages | 19 Pages |
| File Size | 1.63 MB |
| Views | 1723 |
| Downloads | 0 |
| Price: |
Buy Now
|
Description
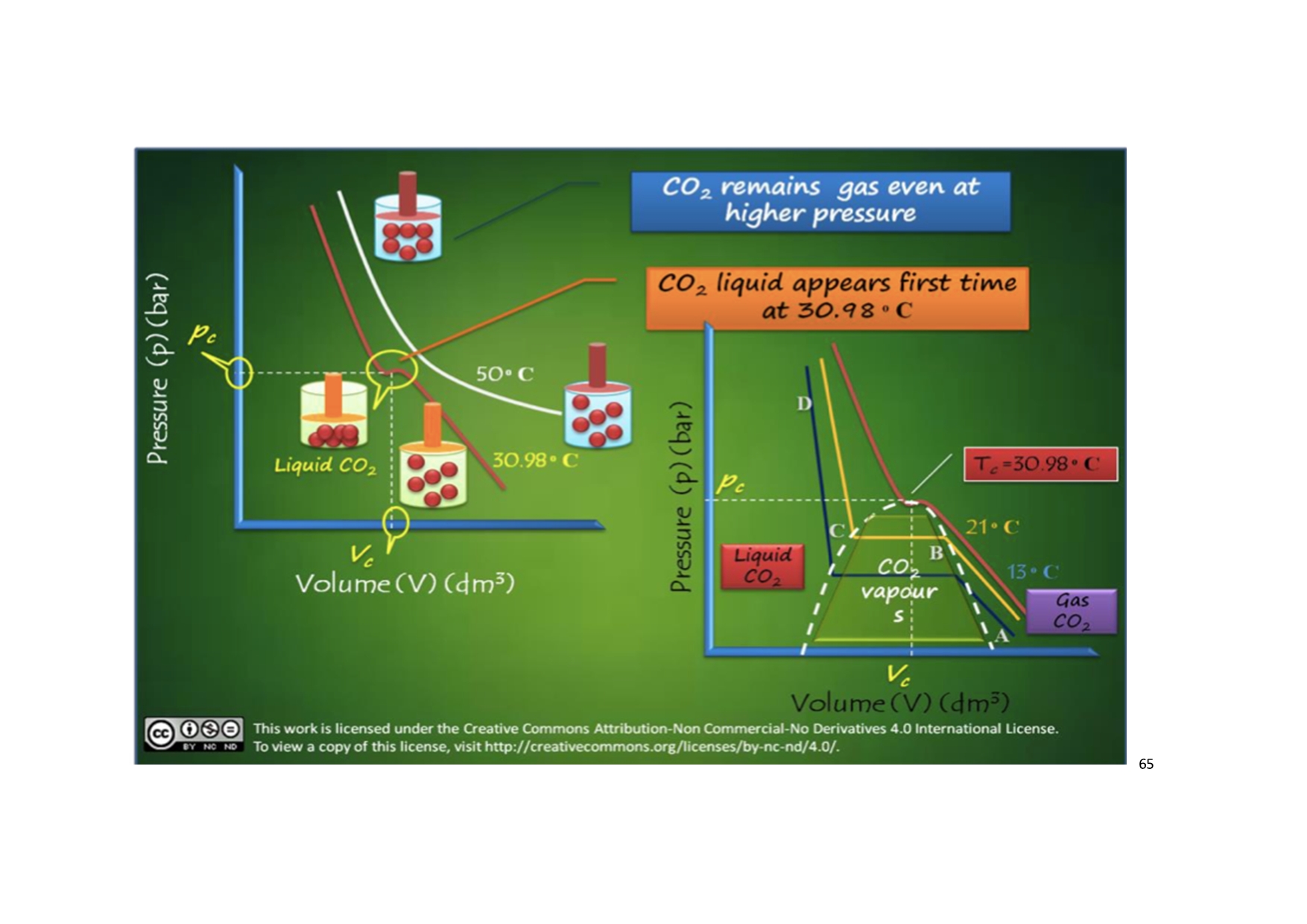
Liquefaction of Gases – Critical Phenomenon
A gas can be liquefied by lowering the temperature and increasing the pressure. At lower temperature, the gas molecules lose kinetic energy. The slow moving molecules then aggregate due to intermolecular attractions between them and are converted into liquid. The increase of pressure - The gas molecules come closer by compression and coalesce to form the liquid.
Below is the document preview.
KCSE FORM 4 CHEMISTRY NOTES
UPDATED KCSE FORM4 CHEMISTRY NOTES
334 Pages
1387 Views
1 Downloads
4.44 MB
KCSE KISWAHILI FORM 1 NOTES
Trending!
UPDATED KCSE KISWAHILI FORM 1 NOTES
23 Pages
2930 Views
3 Downloads
411.07 KB