SPH 302 LESSON 8: Equillibrium between phases
| Institution | TOM MBOYA UNIVERSITY |
| Course | EDUCATION |
| Year | 3rd Year |
| Semester | Unknown |
| Posted By | Brian Mike |
| File Type | |
| Pages | 9 Pages |
| File Size | 410.04 KB |
| Views | 5353 |
| Downloads | 0 |
| Price: |
Buy Now
|
Description
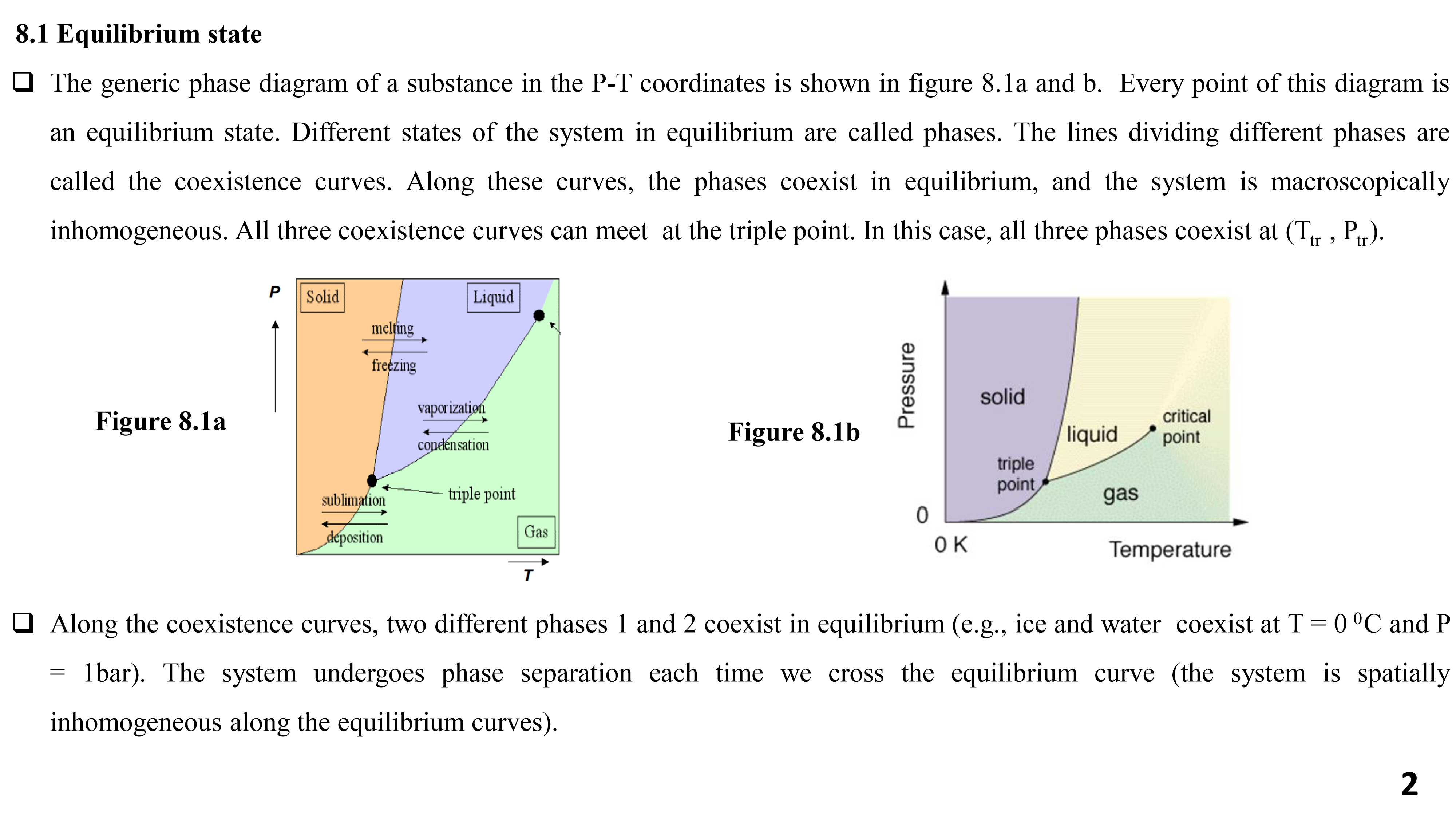
The generic phase diagram of a substance in the P-T coordinates is shown in figure 8.1a and b. Every point of this diagram is an equilibrium state. Different states of the system in equilibrium are called phases. The lines dividing different phases are called the coexistence curves. Along these curves, the phases coexist in equilibrium, and the system is macroscopically inhomogeneous. All three coexistence curves can meet at the triple point.
Below is the document preview.
PHYSICS SCHEMES OF WORK FORM 4
Trending!
UPDATED PHYSICS SCHEMES OF WORK FORM 4
11 Pages
5341 Views
0 Downloads
519.22 KB
BUSSINESS SCHEMES OF WORK FORM 2
Trending!
UPDATED BUSSINESS SCHEMES OF WORK FORM 2
14 Pages
5757 Views
1 Downloads
721.28 KB
BUSSINESS SCHEMES OF WORK FORM 3
Trending!
UPDATED NOTES ON BUSSINESS SCHEMES OF WORK FORM 3
43 Pages
5522 Views
0 Downloads
1.22 MB
FORM 3 ENGLISH NOTES
Trending!
UPDATED FORM 3 ENGLISH NOTES
258 Pages
5239 Views
1 Downloads
1.27 MB
KCSE FORM 1 CHEMISTRY NOTES
Trending!
UPDATED KCSE FORM1 CHEMISTRY NOTES
117 Pages
4677 Views
1 Downloads
2.43 MB
KCSE FORM 2 CHEMISTRY NOTES
Trending!
UPDATED KCSE FORM2 CHEMISTRY NOTES
137 Pages
2298 Views
0 Downloads
1.73 MB
KCSE FORM 3 CHEMISTRY NOTES
Trending!
UPDATED KCSE FORM3 CHEMISTRY NOTES
262 Pages
3071 Views
1 Downloads
2.11 MB