SPH 302 LESSON 8: Equillibrium between phases
| Institution | TOM MBOYA UNIVERSITY |
| Course | EDUCATION |
| Year | 3rd Year |
| Semester | Unknown |
| Posted By | Brian Mike |
| File Type | |
| Pages | 9 Pages |
| File Size | 410.04 KB |
| Views | 5828 |
| Downloads | 0 |
| Price: |
Buy Now
|
Description
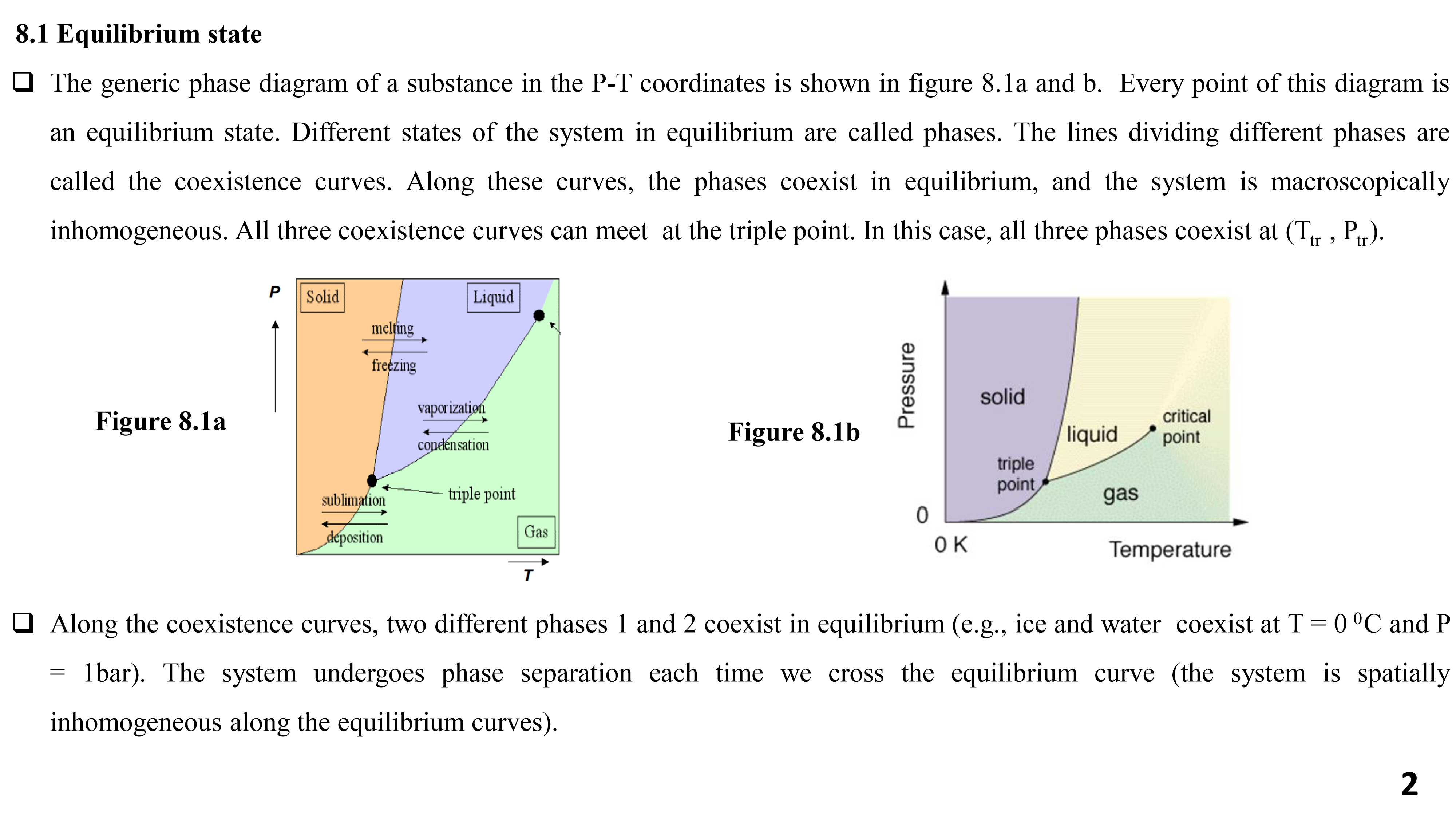
The generic phase diagram of a substance in the P-T coordinates is shown in figure 8.1a and b. Every point of this diagram is an equilibrium state. Different states of the system in equilibrium are called phases. The lines dividing different phases are called the coexistence curves. Along these curves, the phases coexist in equilibrium, and the system is macroscopically inhomogeneous. All three coexistence curves can meet at the triple point.
Below is the document preview.
EET 100 Introduction to Microeconomics
Trending!
Simple and Clear
139 Pages
3859 Views
2 Downloads
1.97 MB
BMS 404 Econometric Models and Methods
Trending!
Simple and Clear
58 Pages
4521 Views
1 Downloads
1.25 MB
SPA 2412; Financial Risk Management Notes 4th Year
Trending!
Financial risk management is a process to deal with key factors affecting interest rates,exchange rates and commodity prices.
99 Pages
4370 Views
3 Downloads
1.31 MB
SPA 2403; Survival Analysis Notes 4th year
Trending!
These are simplified survival analysis notes to help the student to pass his or her exams.
67 Pages
3807 Views
1 Downloads
184.36 KB
Curriculum development notes 2nd year
Trending!
Simple and clear notes
51 Pages
2983 Views
2 Downloads
565.08 KB