ACH 101: Introduction to Inorganic Chemistry
| Institution | UNIVERSITY |
| Course | BIOCHEMISTRY |
| Year | 1st Year |
| Semester | Unknown |
| Posted By | Brian Mike |
| File Type | |
| Pages | 23 Pages |
| File Size | 1.52 MB |
| Views | 3030 |
| Downloads | 0 |
| Price: |
Buy Now
|
Description
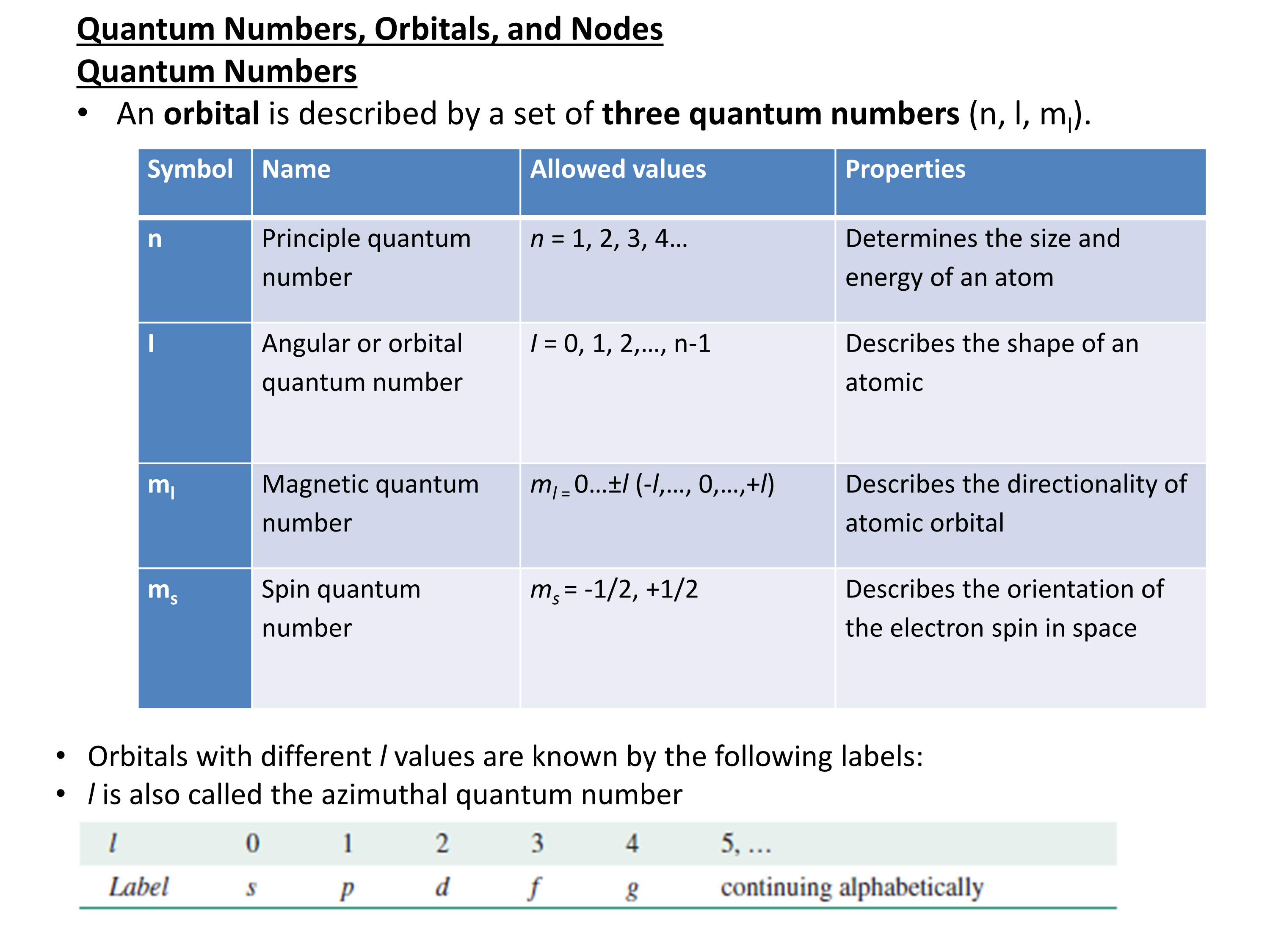
The electronic configuration of cations is assigned by removing electrons first in the outermost p orbital, followed by the s orbital and finally the d orbitals (if any more electrons need to be removed).
Example:
calcium (Z=20), 1s22s22p63s23p64s2; Ca2+: EC is 1s22s22p63s23p6
chlorine (Z=17), EC is 1s22s22p63s23p5 Cl- : 1s22s22p63s23p6.
The ns electrons are always lost before the (n-1)d when forming cations for transition metals. For example,
Zn: [Ar]4s23d10 Zn+2: [Ar]3d10
Below is the document preview.
COMPUTER FORM 1 NOTES
Trending!
UPDATED NOTES ON COMPUTER STUDIES
95 Pages
2582 Views
0 Downloads
1.44 MB
COMPUTER FORM2 NOTES
Trending!
UPDATED NOTES ON COMPUTER STUDIES FORM2
139 Pages
3038 Views
1 Downloads
2.61 MB
COMPUTER FORM1-4 NOTES
Trending!
UPDATED NOTES ON COMPUTER FORM1-4
20 Pages
3118 Views
1 Downloads
649.32 KB
CHEMISTRY NOTES FORM 1-4
Trending!
UPDATED CHEMISTRY NOTES FORM1-4
806 Pages
3532 Views
15 Downloads
9.24 MB
BIOLOGY NOTES FORM 1-4
Recommended!
Trending!
UPDATED BIOLOGY NOTES FORM 1-4
412 Pages
13173 Views
42 Downloads
15.04 MB
GEOGRAPHY NOTES FORM 1-4
UPDATED NOTES ON GEOGRAPHY NOTES FORM 1-4
468 Pages
457 Views
2 Downloads
4.85 MB